|
One bar is 100,000 Pa, and for most practical purposes can be approximated to one atmosphere even if The bar (bar) is commonly used in the industry. Since atmospheric pressure is 14.696 psi - a column of air on a area of one square inch area from the Earth's surface to the space - weights 14.696 pounds. but is now replaced in almost every country except in the US by SI units. Pounds per square inch (psi) was commonly used in the U.K. 10 GPa - pressure enough to form diamondsĪ torr (often used in vacuum applications) is named after Torricelli and is the pressure produced by a column of mercury 1 mm high - equals to 1 / 760 th of an atmosphere.10 MPa - nozzle pressure in a "high pressure" washer.10 kPa - the pressure below 1 m of water, or the drop in air pressure when moving from sea level to 1000 m elevation.1 kPa - approximately the pressure exerted by a 10 g of mass on a 1 cm 2 area.10 Pa - the pressure below 1 mm of water.1 hectoPascal = 100 Pascal = 1 millibar.The unit kiloPascal (kPa) is commonly used in the design of technical applications - like HVAC systems, piping systems and similar. Since 1 Pa is a small pressure unit the unit hectoPascal (hPa) is widely used, especially in meteorology. In imperial units the Standard Atmospheric Pressure is 14.696 psi. The temperature of 293 oK (20 oC) is sometimes used. The Standard Atmospheric Pressure is defined at sea-level at 273 oK (0 oC) and is 1.01325 bar or 101325 Pa (absolute). The Standard Atmospheric Pressure ( atm) is normally used as the reference when listing gas densities and volumes. The atmospheric pressure varies with temperature and altitude above sea level. This pressure is often called the gauge pressure and can be expressed asĪtmospheric pressure is the pressure in the surrounding air at - or "close" to - the surface of the earth. Gauge PressureĪ gauge is often used to measure the pressure difference between a system and the surrounding atmosphere. All calculations involving the gas law requires pressure (and temperature) to be in absolute units. The absolute pressure - p abs - is measured relative to the absolute zero pressure - the pressure that would occur at absolute vacuum. The basic unit for mass is slug and the unit for force is pound ( lb) or pound force ( lb f). P = pressure (lb/in 2 (psi), lb/ft 2 (psf), N/m 2, kg/ms 2 (Pa))ġ) In the Imperial - English Engineering System special care must be taken for the force unit. The equation for pressure can be expressed as:  
So, to convert directly from bar to pounds per square inch, you multiply by 14.503774."the normal force per unit area exerted on a imaginary or real plane surface in a fluid or a gas" Or, you can find the single factor you need by dividing the A factor by the B factor.įor example, to convert from bar to pounds per square inch you would multiply by 100000 then divide by 6894.757. 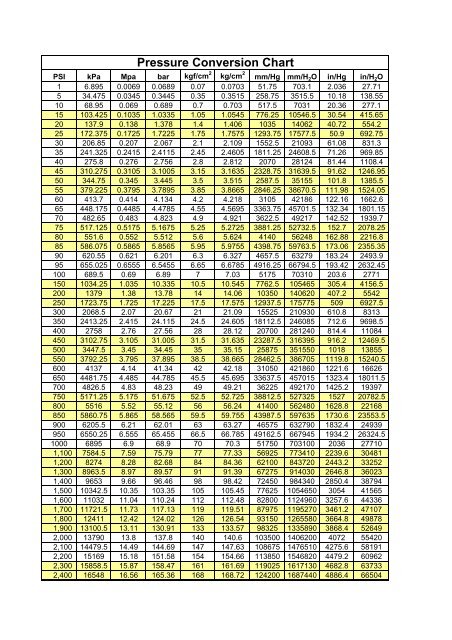
To convert among any units in the left column, say from A to B, you can multiply by the factor for A to convert A into Pascals then divide by the factor for B to convert out of Pascals. To convert from Pa into units in the left columnĭivide by the value in the right column or, multiply by the reciprocal, 1/x.ĥ00000 Pa / 100000 = 5 bar 
Multiply by the conversion value in the right column in the table below.ĥ bar * 100000 = 500000 Pa To simply convert from any unit into pascals, for example, from 5 bar, just Where S is our starting value, C is our conversion factor, and How to Convert Units of PressureĬonversions are performed by using a conversion factor. By knowing the conversion factor, converting between units can become a simple multiplication problem: Some are obvious, such as pounds per square inch, but even the SI standard Pascal is actually an expression of one Newton per square meter. Since pressure is derived from force and area, many units for pressure directly relate force to area. Where P is equal to pressure, F is the normal force and A is area. Pressure is a scalar quality reflecting how force acts on a surface.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
